Novel Internalin P homologs in Listeria ivanovii londoniensis and Listeria seeligeri
Abstract
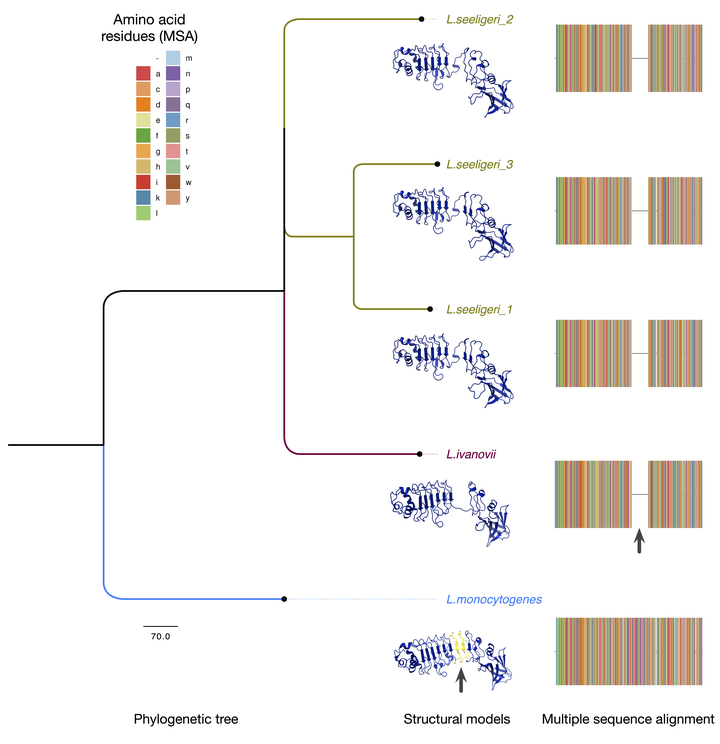
Listeria monocytogenes (Lm) is a bacterial pathogen that causes listeriosis in immunocompromised individuals, particularly pregnant women. Several virulence factors support the intracellular lifecycle of Lm and facilitate cell-to-cell spread, allowing it to occupy multiple niches within the host and cross protective barriers, including the placenta. One family of virulence factors, internalins, contributes to Lm pathogenicity by inducing specific uptake and conferring tissue tropism. Over 25 internalins have been identified thus far, but only a few have been extensively studied. Internalins contain leucine-rich repeat (LRR) domains which enable protein-protein interactions, allowing Lm to bind host proteins. Notably, other Listeria species express internalins but cannot colonize human hosts, prompting questions regarding the evolution of internalins within the genus Listeria. Internalin P (InlP) promotes placental colonization through interaction with the host protein afadin. Though prior studies of InlP have begun to elucidate its role in Lm pathogenesis, there remains a lack of information regarding homologs in other Listeria species. Here, we have used a computational evolutionary approach to identify InlP homologs in additional Listeria species. We found that L. ivanovii londoniensis (Liv) and L. seeligeri (Ls) encode InlP homologs. We also found InlP-like homologs in L. innocua and the recently identified species L. costaricensis. All newly identified homologs lack the full-length LRR6 and LRR7 domains found in Lm’s InlP. These findings inform on the evolution of one key Lm virulence factor, InlP, and serve as a springboard for future evolutionary studies of Lm pathogenesis as well as mechanistic studies of Listeria internalins.
KNC and JTB are co-primary authors. JR and JWH are co-corresponding authors.